Slag constitutes a vital phase in various metallurgical reactions, serving both to remove impurities from steel and to protect molten steel. Maintaining optimal slag conditions throughout all stages of electric furnace steelmaking is one of the key prerequisites for producing high-quality steel.

I. Sources of Slag
Throughout the various stages of electric arc furnace steelmaking, molten steel surfaces are covered by slag originating from four primary sources:
1) Deliberately added slag-forming materials to achieve smelting objectives, such as lime, limestone, silica sand, fluorspar, iron ore, carbon powder, ferrosilicon powder, and refractory brick fragments.
2) Products formed during smelting, primarily oxides or sulphides of various elements present in the steel, such as FeO, MnO, SiO₂, Cr₂O₃, etc.
3) Refractory materials eroded from the furnace lining and incorporated into the slag, such as MgO in basic slag and SiO₂ in acidic slag.
4) Impurities introduced by various raw materials.
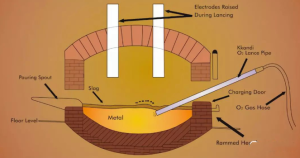
II. Classification and Composition of Slag
During steelmaking, slag constitutes a crucial phase participating in various metallurgical reactions. Consequently, different steelmaking processes and distinct smelting stages employ different types of slag.
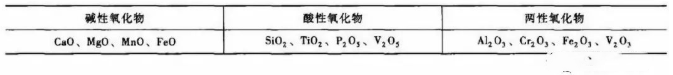
1) Electric arc furnace linings are categorised as either basic or acidic, resulting in corresponding basic or acidic slags. Slags dominated by basic oxides are termed basic slags; those dominated by acidic oxides are termed acidic slags. Common oxides in slags, classified by their acidic or basic properties, are presented in Table 1.
Table 1 Classification of Common Oxides in Slags
Alkalinity is a parameter characterising the acidic or basic properties of slag. Theoretically, it should represent the ratio of the sum of the mass fractions of various basic oxides to the sum of the mass fractions of various acidic oxides. In actual production, alkalinity is commonly expressed as the ratio of the mass fraction of CaO to the mass fraction of SiO₂. Slag with an alkalinity greater than 1 is classified as basic slag, while that with an alkalinity less than 1 is classified as acidic slag.
2) By smelting stage, slag may be categorised as melting slag, oxidising slag, or reducing slag. Slag from the reduction phase of an alkaline electric arc furnace may further be classified as white slag or calcium carbide slag.
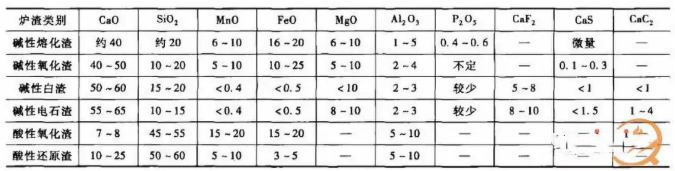
3) The physical and chemical properties of slag are determined by its composition. For steelmaking slag, the most critical physical property is viscosity; chemically, the key attributes are alkalinity and the strength of its oxidising (or reducing) character. Slag primarily consists of various oxides, such as SiO₂, CaO, Al₂O₃, MgO, FeO, MnO, and P₂O₅. Additionally, it contains minor sulphides like CaS, FeS, and MnS. The approximate composition of different slags during electric arc furnace steelmaking is shown in Table 2.
Table 2 Approximate Composition of Various Slags (Mass Fraction)
III. The Role of Slag
During steelmaking, slag serves the following functions:
1) Controlling oxidation and reduction of molten steel. The primary stages of steelmaking involve oxidation and reduction. During oxidation, certain elements in the molten steel must be oxidised; during reduction, residual oxygen must be removed. Controlling oxygen content in the molten steel necessitates regulating the FeO content within the slag.
2) Phosphorus and sulphur are both harmful impurities in steel. Their removal relies on a properly composed slag with a strong affinity for oxidised phosphorus or sulphides, enabling their transfer from the molten steel into the slag.
3) The high temperature of the arc column enables gases to dissociate into ionic states for absorption by the molten steel. The slag layer covering the molten steel surface provides excellent protection, reducing gas ingress. The slag’s elevated temperature and low thermal conductivity minimise heat loss from the molten steel, maintaining temperature stability.
4) Protection of the furnace lining. The slag layer envelops the arc, reducing its radiative erosion of the lining; this effect is enhanced when producing foamed slag.
5) Removal of non-metallic inclusions. Steelmaking charge materials often contain impurities such as sand and soil, oxides formed from various elements, and damaged or exfoliated refractory lining materials. If these enter the steel, they become non-metallic inclusions. Such non-metallic inclusions possess high melting points—e.g., SiO₂ at 1710°C and Al₂O₃ at 2050°C—and remain unmelted at steelmaking temperatures. However, when combined with slag to form composite compounds, their melting points decrease significantly. These compounds become liquid, readily coagulate, and separate from the molten steel.
IV. Viscosity of Slag
Viscosity is an indicator reflecting the viscous resistance to fluid flow. During laminar flow, friction resistance exists between successive fluid layers, impeding movement. This internal friction resistance constitutes one of a fluid’s inherent properties. Viscosity is inversely proportional to fluidity; higher viscosity indicates poorer flowability. Viscosity is broadly categorised into three types: dynamic viscosity, kinematic viscosity, and conditional viscosity. Unless otherwise specified, references to ‘viscosity’ typically denote dynamic viscosity.
The frictional resistance generated when successive fluid layers move relative to one another is proportional to both the contact area between these layers and the relative velocity of their motion. The proportional constant between these factors constitutes the dynamic viscosity of the liquid, commonly denoted by the symbol η and measured in pascals per second (Pa·s). The kinematic viscosity of a liquid is obtained by dividing its dynamic viscosity by its density. Conditional viscosity refers to viscosity measurements taken using various calibrated viscometers, such as the Engler viscometer or the cup viscometer.
Viscosity is one of the crucial physical properties of slag, significantly influencing various reactions between molten steel and slag, the escape of gases from steel, heat transfer, and even the service life of furnace linings. The efficiency of dephosphorisation and desulphurisation, as well as the rates of oxidation and reduction reactions, depend on the diffusion speed of reactants within the slag.
Excessively high slag viscosity hinders numerous metallurgical reactions between molten steel and slag. For instance, during decarburisation in the oxidation phase, substantial resistance to CO bubble escape through the slag slows the carbon-oxygen reaction. Similarly, diffusion deoxidation is significantly impaired during reduction. The same applies to phosphorus and sulphur removal.
Excessively low slag viscosity is also detrimental. If the slag is too fluid, it struggles to absorb heat from the arc column, increasing the portion reflected onto the furnace roof and walls. This hinders temperature control of the molten steel and shortens the refractory lining’s lifespan. Simultaneously, low-viscosity, highly fluid slag exerts intense direct erosion and scouring effects on the lining.
In practical production, maintaining stable slag viscosity is of paramount importance.
In basic slags, adding acidic oxides reduces viscosity, while alkaline oxides increase it. The opposite holds true for acidic slags. However, exceptions exist: although FeO is an alkaline oxide, it lowers the viscosity of basic slags due to its low melting point.